Ultraviolet-Visible Spectrometry
- Absorption of visible and Ultraviolet radiation is associated with the excitation of electrons.
- The electrons jump from the lower levels to higher levels.
- Only light at a specific wavelength can be absorbed so it is possible to identify the molecule.
Ultraviolet Instrument
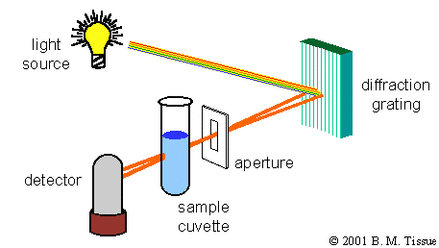
- The light source is directed towards a diffraction grating where the light is directed towards the sample.
- The specific wavelength of choice is selected and shone directly through the sample.
- The specific wavelength is absorbed and the detector analyses the intensity of the light that reaches it relative to the light that was passed through the sample.
- The detector translates this information and produces a spectrum.
- The absorption is directly proportional to the concentration of the sample.
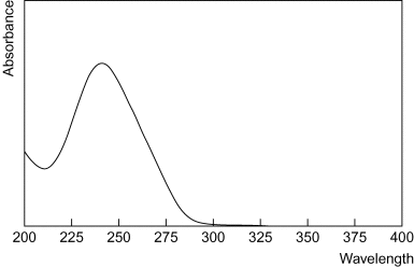
Ultraviolet Spectrum
Uses of Ultraviolet Spectrometry
- UV-Visible spectrometry is a technique that readily allows for the determination of concentration of molecules.
- It can be used for the quantification of DNA, proteins and enzymes.
- In the environmental industry UV-Visible spectrometry is used for the quantification of organic materials and heavy metals present in water.
- In clinical chemistry UV-Visible spectrometry is used in the study of enzyme kinetics. When cells are damaged by disease they leak enzymes into the blood stream. The concentration of enzymes in the blood can determine the severity of the disease.